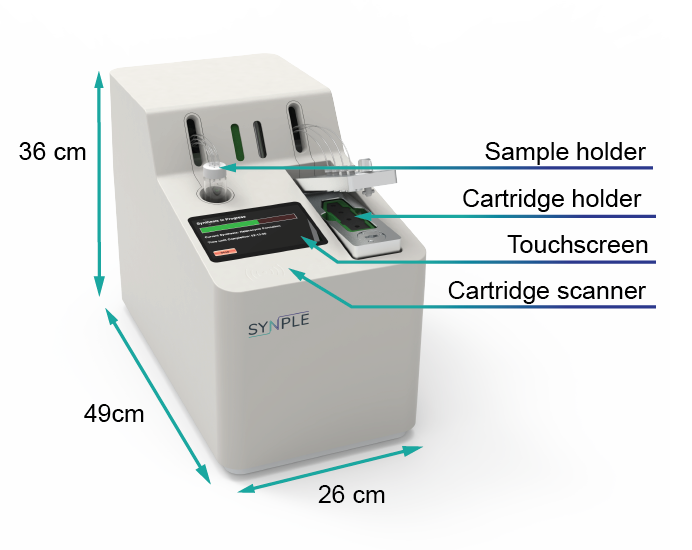
Synple 2

The 3 in 1 solution offers an unprecedented combination of hardware, software and an optimized chemical process. Use of the automated synthesizer, in conjunction with the specialised capsules, offers the opportunity to break free of the limitations imposed by traditional synthesis, and the benefits of a safe, time-efficient, cost effective, plug and play synthetic procedure.
How can Synple help me?
The first automated synthesis device to support your daily work
The Synple 2 is the first easy to use automated synthesis device that increases your daliy productivity without any hassles.
Be in control! – Flexibility to choose from a range of reactions and modify parameters
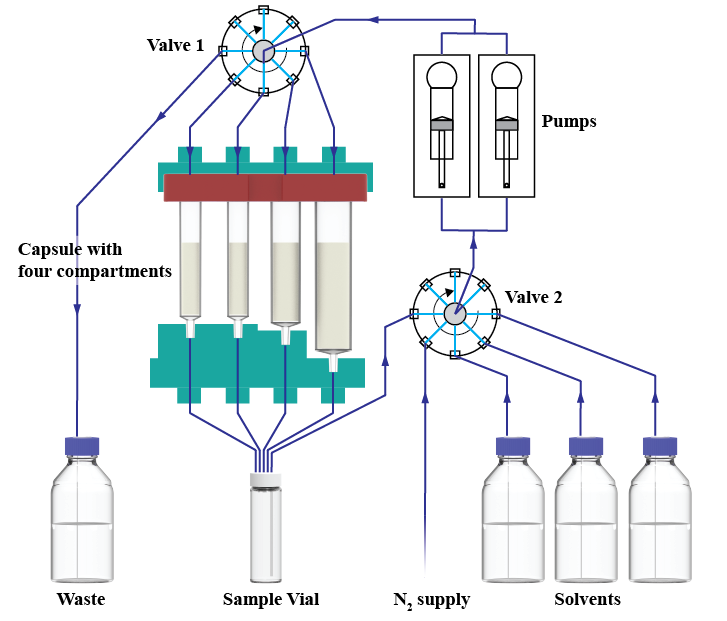
- Reagent cartridges are available for eight distinct reaction classes, with more than different 40 reagents.
- Each cartridge contains a scannable chip enabling the the standardized reaction sequence to be loaded without any further programming.
- Key reaction parameters (time, temperature, etc.) can be easily modified.
Click on the cartridge to see what reagents are contained in them and how the reactions work. A detailed set of application notes is available for each type of cartridge
Work smarter – With Synple you can focus on the experiments that matter
Synple allows you to run your daily experiments in a standardized, completely automated manner, giving you more time to think and focus on the more demanding experiments.
The video illustrates how much time is actually required to run the same experiments manually vs on the Synple instrument.
With Synple you can now do the same work in 5-10 minutes, which otherwise takes several hours.
Deliver Faster – Get more done in less time
Synple offers standardized, “off the shelf” reactions and, with the support of our clear reaction scope, the confidence to know that you will get your product at the end without having to spend time optimizing reactions.
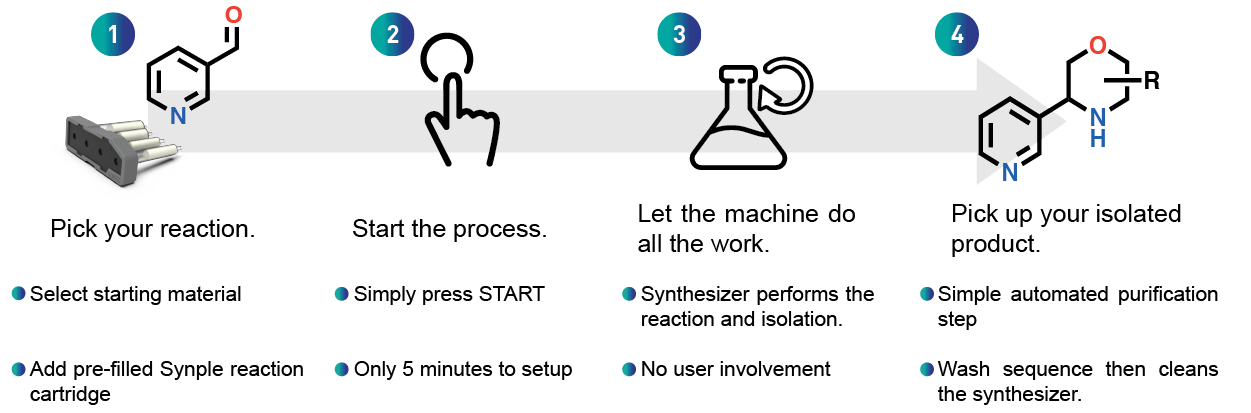
In just 4 easy and quick steps you can set up the whole reaction and then have time to focus on other work or reactions.
At the end of each day you can make more compounds than you would have without the Synple 2.
How our customers use Synple
How to work with the Synple 2
Fast and easy reaction setup
Automated cleaning sequence
Advantages of Synple

5x more efficient

No training required - Easy to use
The plug&play device is intuitive to use and requires no special training. No detailed chemistry knowledge is necessary to create new molecules.

70% Lower costs
Compared to carrying out a reaction manually, the synthesizer can cut down on 70% of the costs to generate one molecule.

High Safety
The automatic reactor is fully enclosed, thus the user has no exposure to any chemicals used during the synthetic process. This complies with newest laboratory safety standards.

Small footprint

90% less waste
The fully optimized reaction sequence uses only a minimum amount of reagents and solvents, therefore reducing overall waste.
Frequently asked questions (FAQ)
Who is Synple Chem?
How many products are on the market?
Can I put my own materials in the cartridges?
What solvents are compatible with the machine?
How does the machine handle poorly soluble starting materials?
How are lower reacting starting materials handled?
Can I run more than one reaction at the same time?
How much maintenance is required?
Do you have products on the market?
In which countries is Synple available?
What is the lead time for the machine and cartridges?
Can my starting material be dissolved in a broad range of solvents?
Does the machine allow for reaction monitoring?
Can the machine be connected to analytical or purification systems?
How long does a run take?
A detailed overview can be found in the ressource section.
Testimonials from Synple Users
As a beta-tester I have successfully used Synple’s technology for all its existing applications. The N-heterocycle forming application is easy to use and I would much rather use this machine to do SnAP chemistry then do the reaction in a flask. The reductive aminations also work well and the PROTAC application is a great way forward for this technology!
The N-heterocycle forming application is easy to use and I would much rather use this machine to do SnAP chemistry then do the reaction in a flask